Usually, non-metals have 5, 6, or 7 electrons in their valence shells. The gain of electrons is termed as formation of anions. If an element has 5, 6, or 7 electrons in its outermost shell, in that case, it tends to gain them to attain the stable configuration or octet. Therefore, the metals lose electrons to attain positively charged ions or cations.įor the sample, potassium, sodium, calcium, magnesium, aluminum, etc., are all metals that can donate their valence electrons to attain a cation state. Usually, metals have 3, 1, or 2 electrons in their valence shells. The loss of electrons is termed as formation of cations. If an element has 1, 3, or 2 electrons in its outermost shell, in that case, it tends to lose them to attain the stable configuration or octet. /GettyImages-604346724-589a56263df78caebc80b0c5.jpg)
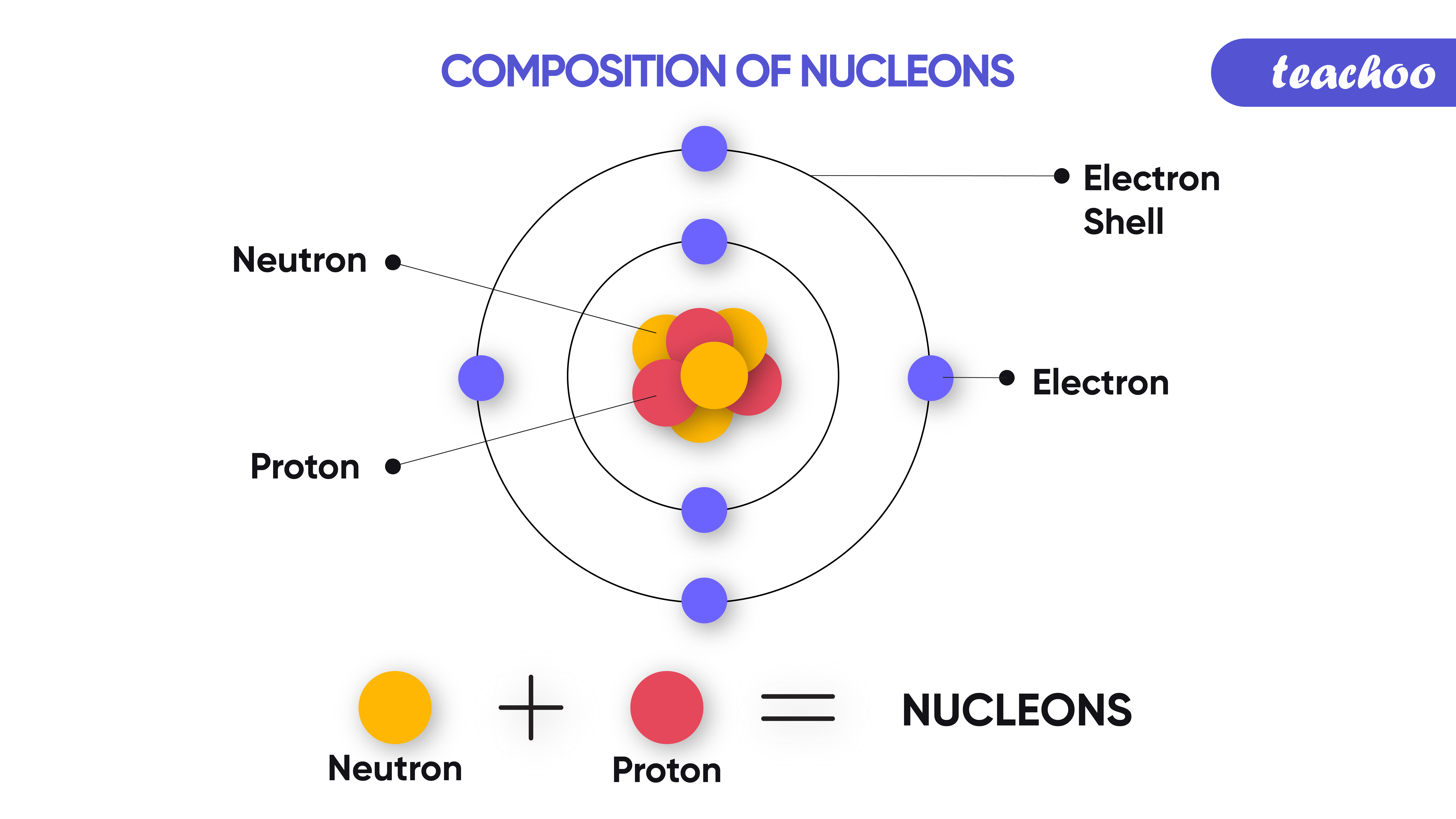
After that, these ions are deposited on the cathode and anode. This method passes electricity through certain conducting solutions, forming ions in the solution. When an ionic compound, like salt, is dissolved in an appropriate solvent, water, etc., the salt atoms dissociate, forming Na⁺ (sodium ion) and Cl⁻ (chloride ion) free ions. The free electron may connect to another molecule or atom, forming a new negatively charged anion. As a result, cations and anions are formed. When molecules of liquid or gaseous fluids collide spontaneously, electrons drop out from one of the atoms or molecules. There are some ways to form ions in an atom. In other words, an anion contains more electrons than protons. Since an anion is devised by adding one or more electrons to an atom, it means an anion has more electrons compared to a normal atom. For instance, a chlorine atom gains one electron to form a chloride ion, Cl⁻, which is an anion. An anion is devised by an atom’s gain of one or more electrons. An Anion or a Negatively Charged Ion:įluoride ions, F⁻, or sulfide ions, S²⁻, are called anions because they are negatively charged ions. In other words, a cation contains fewer electrons than protons. For instance, a sodium atom drops one electron to form a sodium ion, Na⁺, which is a cation.Īs the cation is formed by removing one or more electrons from an atom, it means a cation has less number of electrons compared to its atom. A cation is devised by the loss of one or more electrons by an atom. A Cation or Positively Charged Ion:Ĭations are called cations if an ion contains a positive charge with some magnitude, such as copper ion, Cu²⁺, etc. There are two kinds of ions: cations and anions. For instance, as the charge on sodium ion is +1, you can write it as Na⁺. If the magnitude of the charge has a value equal to one (either + or -), you can omit the magnitude of the charge. For instance, the oxide ion is represented as O²⁻. If the magnitude is greater than -1, the charge of magnitude is written after the magnitude. Now, the magnitude of the charge on an ion is written in the superscript format next to the chemical formula of the ion.Īfter writing magnitude, move to the charge or sign of the magnitude, i.e., positive (+) or negative (-). It is necessary to write the chemical formula of an atom before representing it in a charged ion form. Later, in 1884, Svante August Arrhenius further proposed the mechanism of ions in his experiment. He described ions as electrically charged atoms or a family of atoms traveling toward a cathode or anode. The word ion is acquired from the Greek work ἰόν, which means ‘to go.’ Therefore, the term ‘ion’ implies ‘a goer.’ In 1830, ions were first introduced by Micheal Faraday. If two or more different atoms form an ion, they are called molecular or polyatomic ions. If an ion is formed by only one variety of atoms (each holding some gross charge, either positive or negative), in that case, the group of atoms can be referred to as a monatomic ion or an atomic ion. Sodium ion (Na⁺), magnesium ion (Mg²⁺), chloride ion (Cl⁻), and oxide ion (O²⁻). Therefore, it contains an unequal number of electrons and protons. Ions DefinitionĪn ion is formed by an atom’s loss or gain of electrons. In simple words, an ion or ions definition can be described as an electrically charged atom or group of atoms. As a universally acknowledged fact, ions of opposite charges always attract each other. The atoms that tend to form cations are generally known as metals, while those that tend to form anions are called non-metals. An ion with a total positive charge is termed a proton, while an ion with a net negative charge is termed an electron.Ī proton is also known as a cation, whereas you can call an electron an anion. It is an atom, group of atoms, or any subatomic particle with a net electric charge. Ions are chemically charged species that contain either a positive or a negatively charged quantity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
